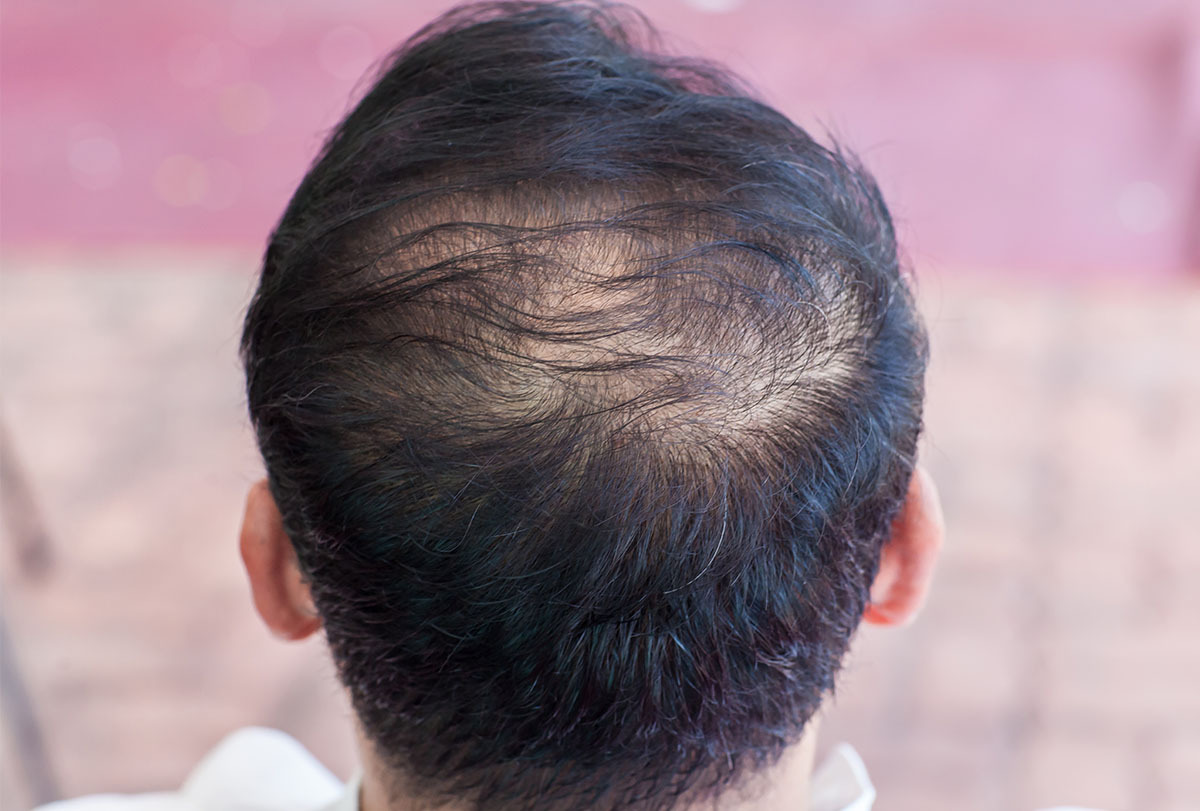
South Korea’s JW Pharmaceutical has received a patent in Russia for a hair loss treatment currently under development, marking the first time that its candidate drug has been patented overseas.
The pharmaceutical firm received a patent for JW0061, its Wnt-targeted hair loss treatment, from Russia's Federal Service for Intellectual Property.
The patent covers a new heterocyclic inducer of the Wnt signaling pathway, including its base or isomer, and compositions containing it as an active ingredient.
JW has also submitted patent applications in more than ten nations, including the US, EU, Japan, and China.
JW0061 exhibits a novel method of action by directly binding to the GFRA1 protein in human hair dermal papilla cells, activating the Wnt signaling pathway. This is the first instance of a low-molecular-weight medication that describes the mechanism of hair growth that has been documented.
JW is now evaluating non-clinical toxicity in accordance with good laboratory practice with the intention of initiating clinical trials for JW0061 in the first half of 2024. Additionally, it collaborates on research projects with eminent dermatologists in the US.
JW plans to develop JW0061 as a new innovative drug that supplements or replaces existing hair loss treatments.



 Goldman Sachs Delays Fed Rate Cut Forecast to 2026 Amid Rising Inflation Concerns
Goldman Sachs Delays Fed Rate Cut Forecast to 2026 Amid Rising Inflation Concerns  Aker BP Q1 Profit Jumps on Higher Oil Prices and Asset Reversal
Aker BP Q1 Profit Jumps on Higher Oil Prices and Asset Reversal  Cogent Biosciences Soars 120% on Breakthrough Phase 3 Results for Bezuclastinib in GIST Treatment
Cogent Biosciences Soars 120% on Breakthrough Phase 3 Results for Bezuclastinib in GIST Treatment  Trump Signs Executive Order to Boost AI Research in Childhood Cancer
Trump Signs Executive Order to Boost AI Research in Childhood Cancer  NASA Resumes Cygnus XL Cargo Docking with Space Station After Software Fix
NASA Resumes Cygnus XL Cargo Docking with Space Station After Software Fix  US Auto Industry Urges Trump to Block Chinese EV Market Access
US Auto Industry Urges Trump to Block Chinese EV Market Access  Novocure Stock Surges 30% After FDA Approves Optune Pax for Pancreatic Cancer Treatment
Novocure Stock Surges 30% After FDA Approves Optune Pax for Pancreatic Cancer Treatment  SpaceX Prioritizes Moon Mission Before Mars as Starship Development Accelerates
SpaceX Prioritizes Moon Mission Before Mars as Starship Development Accelerates  Is space worth the cost? Accounting experts say its value can’t be found in spreadsheets
Is space worth the cost? Accounting experts say its value can’t be found in spreadsheets  Trump to Visit China for Key U.S.-China Summit With Xi Jinping
Trump to Visit China for Key U.S.-China Summit With Xi Jinping  Intermittent fasting doesn’t have an edge for weight loss, but might still work for some
Intermittent fasting doesn’t have an edge for weight loss, but might still work for some  US Stock Futures Slip as Trump Rejects Iran Peace Proposal Amid Rising Middle East Tensions
US Stock Futures Slip as Trump Rejects Iran Peace Proposal Amid Rising Middle East Tensions  Eli Lilly’s Inluriyo Gains FDA Approval for Advanced Breast Cancer Treatment
Eli Lilly’s Inluriyo Gains FDA Approval for Advanced Breast Cancer Treatment  CSL Shares Crash as Profit Warning and $5 Billion Impairment Shake Investors
CSL Shares Crash as Profit Warning and $5 Billion Impairment Shake Investors