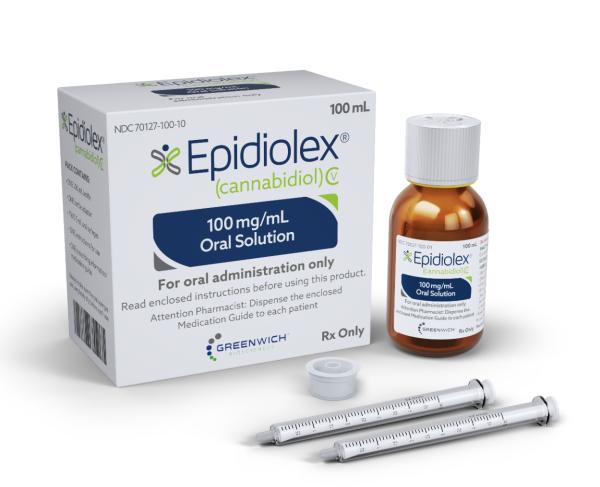
The US Food and Drug Administration (FDA) is planning to regulate the use of the cannabis compound CBD in food and supplements after weighing the evidence on the compound's safety.
The FDA will decide whether regulating legal cannabis will require new agency rules or new legislation from Congress.
Cannabis products, excluding Epidiolex, are illegal at the federal level in the US, although some states allow their use.
According to Patrick Cournoyer, who heads the FDA office developing the agency's cannabis strategy, the FDA will ascertain if CBD can be safely eaten every day for a long period or during pregnancy amid concerns about future fertility.
.



 Texas Sues Meta Over WhatsApp Encryption Claims
Texas Sues Meta Over WhatsApp Encryption Claims  Judge Delays SEC Settlement With Elon Musk Over Twitter Stock Disclosure Case
Judge Delays SEC Settlement With Elon Musk Over Twitter Stock Disclosure Case  Fed Signals Possible Rate Hikes if Inflation Remains High in 2026
Fed Signals Possible Rate Hikes if Inflation Remains High in 2026  US Stock Futures Slip as Nvidia Earnings Fail to Fully Impress Investors
US Stock Futures Slip as Nvidia Earnings Fail to Fully Impress Investors  Judge Rules Use of Military Lawyers in Civilian Prosecutions Is Lawful
Judge Rules Use of Military Lawyers in Civilian Prosecutions Is Lawful  CDC Monitors 41 Americans After Andes Hantavirus Outbreak on Luxury Cruise Ship
CDC Monitors 41 Americans After Andes Hantavirus Outbreak on Luxury Cruise Ship  Coles “Down Down” Ruling Sparks Fresh Scrutiny of Australian Supermarket Pricing
Coles “Down Down” Ruling Sparks Fresh Scrutiny of Australian Supermarket Pricing  Minnesota ICE Agent Charged in Venezuelan Immigrant Shooting During Trump Immigration Crackdown
Minnesota ICE Agent Charged in Venezuelan Immigrant Shooting During Trump Immigration Crackdown  Standard Chartered Targets Higher Profitability With Major Workforce Cuts
Standard Chartered Targets Higher Profitability With Major Workforce Cuts  Mistral AI Acquires Emmi AI to Expand Industrial AI Solutions in Europe
Mistral AI Acquires Emmi AI to Expand Industrial AI Solutions in Europe  OpenAI Eyes IPO Filing as Early as This Week Amid Rising AI Competition
OpenAI Eyes IPO Filing as Early as This Week Amid Rising AI Competition  Lam Research Expands AI-Powered Semiconductor Tools and Arizona Operations
Lam Research Expands AI-Powered Semiconductor Tools and Arizona Operations  Asian Stocks Mixed as Tech Shares Slide Ahead of Nvidia Earnings
Asian Stocks Mixed as Tech Shares Slide Ahead of Nvidia Earnings  RFK Jr. Expands CDC Vaccine Advisory Panel's Scope Amid Legal Battles
RFK Jr. Expands CDC Vaccine Advisory Panel's Scope Amid Legal Battles  Trump-Xi Summit Sparks Renewed Hope for Americans Detained in China
Trump-Xi Summit Sparks Renewed Hope for Americans Detained in China  Daiichi Sankyo Stock Drops After Earnings Delay and Oncology Review
Daiichi Sankyo Stock Drops After Earnings Delay and Oncology Review