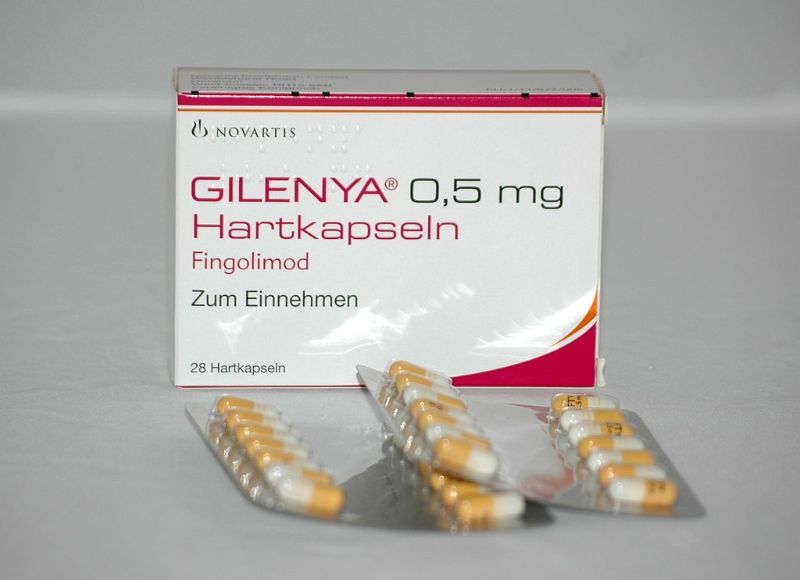
A US district court upheld the validity of Novartis' patent for its top-selling multiple sclerosis (MS) drug Gilenya, rejecting a lawsuit from generic drug maker HEC.
Gilenya is the Swiss drugmaker's No. 2 revenue generator at $738 million in the second quarter.
HEC had sought approval to make a generic copy of Gilenya before the expiration of Novartis' patent with the US Food and Drug Administration (FDA).
The Swiss drugmaker said it continued the injunction against the marketing and sale of Gilenya and other generics granted to it in June 2019.
The FDA approved three generic versions of the Novartis MS treatment in December.
Novartis had settlement agreements with other manufacturers to allow them to launch generic versions of Gilenya before the patent expires in 2027.



 GOP Lawmakers Probe Sam Altman and OpenAI Ahead of Potential IPO
GOP Lawmakers Probe Sam Altman and OpenAI Ahead of Potential IPO  Maersk Q1 Earnings Beat Expectations as Iran Conflict Clouds Shipping Outlook
Maersk Q1 Earnings Beat Expectations as Iran Conflict Clouds Shipping Outlook  Orsted Q1 EBITDA Beats Expectations Despite U.S. Impairments
Orsted Q1 EBITDA Beats Expectations Despite U.S. Impairments  K+S Raises 2026 Earnings Outlook After Strong Q1 Results
K+S Raises 2026 Earnings Outlook After Strong Q1 Results  Kuaishou Stock Jumps on Kling AI IPO Plans and $20 Billion Valuation
Kuaishou Stock Jumps on Kling AI IPO Plans and $20 Billion Valuation  Nike Tariff Refund Lawsuit Sparks Consumer Backlash Over Price Increases
Nike Tariff Refund Lawsuit Sparks Consumer Backlash Over Price Increases  CSL Shares Crash as Profit Warning and $5 Billion Impairment Shake Investors
CSL Shares Crash as Profit Warning and $5 Billion Impairment Shake Investors  AWS Data Center Overheating Disrupts Cloud Services in Northern Virginia
AWS Data Center Overheating Disrupts Cloud Services in Northern Virginia  Aker BP Q1 Profit Jumps on Higher Oil Prices and Asset Reversal
Aker BP Q1 Profit Jumps on Higher Oil Prices and Asset Reversal  Morgan Stanley Bets on Optical Component Stocks in Greater China Tech Sector
Morgan Stanley Bets on Optical Component Stocks in Greater China Tech Sector  Samsung Shares Slide as Labor Talks Intensify Ahead of Planned Strike
Samsung Shares Slide as Labor Talks Intensify Ahead of Planned Strike  Goldman Sachs Delays Fed Rate Cut Forecast to 2026 Amid Rising Inflation Concerns
Goldman Sachs Delays Fed Rate Cut Forecast to 2026 Amid Rising Inflation Concerns  Armani Group Eyes Strategic Stake Sale to Luxury Giants
Armani Group Eyes Strategic Stake Sale to Luxury Giants  Hantavirus Cruise Ship Outbreak Triggers Global Health Alert
Hantavirus Cruise Ship Outbreak Triggers Global Health Alert  AcadeMedia Q3 Profit Climbs as International and Adult Education Segments Drive Growth
AcadeMedia Q3 Profit Climbs as International and Adult Education Segments Drive Growth  US Auto Industry Urges Trump to Block Chinese EV Market Access
US Auto Industry Urges Trump to Block Chinese EV Market Access  Coinbase Q1 2026 Earnings Miss Sends COIN Stock Lower Amid Crypto Market Slump
Coinbase Q1 2026 Earnings Miss Sends COIN Stock Lower Amid Crypto Market Slump